Background
The removal of oxygen from a liquid gas stream is often the most crucial protection against the degradation of equipment or chemicals. Just outside of the CRS lab, we have a bank of AFS I and AFS II purifiers to polish incoming gases such as hydrogen, dry air, and helium from several standard tanks. To scrub the incoming nitrogen, we use various purifiers such as the AFS I and the ZPure DS PolyGas I at the point of use. These purifiers remove oxygen, moisture, and hydrocarbon (we will set aside another blog to discuss moisture and hydrocarbon removal).
The Chemistry
Though the oxygen adsorbents in the AFS I and the ZPure DS PolyGas I purifiers are different, the basic chemical reactions are the same:
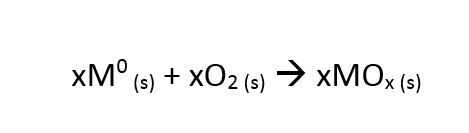
In other words, a transition metal (M) reacts with oxygen (O) to form a metal oxide (MO). The x’s simply denote that the stoichiometry is arbitrary for this example, since it is dictated by the specific oxidation state of the transition metal. The benefit of this type of chemical scrubbing is that no gaseous byproduct is formed.
It’s easy to imagine the reactivity of certain transition metals, such as iron or copper, when you see them rust or tarnish in the air. The slow rate of ambient oxidation would lead you to believe that the reaction rate of our oxygen traps is slow. However, the reaction is accelerated by two factors. First, chemical promoters are added to the adsorbent to lower the activation energy of the active metal surface. Second, the metal surface is incorporated in a highly porous matrix, which increases the surface area on which the oxidation reaction can occur.
The Application
 The reaction of O2 on activated metal adsorbents is diffusion limited, since the gas molecules must travel through the tortuous paths of the porous matrix. Therefore, residence time, or the average time a molecule spends in a gas purifier, plays a key role in determining the oxygen capacity of the adsorbent. Increasing the flow rate of the carrier gas decreases the residence time of the molecules in the purifier, which results in a decrease in total oxygen capacity. In a sense, increasing the flow rate decreases the time that each oxygen molecule interacts with the reactive metal sites.
The reaction of O2 on activated metal adsorbents is diffusion limited, since the gas molecules must travel through the tortuous paths of the porous matrix. Therefore, residence time, or the average time a molecule spends in a gas purifier, plays a key role in determining the oxygen capacity of the adsorbent. Increasing the flow rate of the carrier gas decreases the residence time of the molecules in the purifier, which results in a decrease in total oxygen capacity. In a sense, increasing the flow rate decreases the time that each oxygen molecule interacts with the reactive metal sites.
When we write specs for our O2 traps, we make sure to specify the nominal flow rate for which the O2 capacity is valid. For example, the ZPure XLS PolyGas I Purifier has 1000 cc O2 capacity at a nominal flow rate of 1.85 L/min. At lower flow rates, the capacity is higher and the opposite is true at higher flow rates. For example, using a flow rate of 550 mL/min would yield a whopping 1700 cc O2 capacity. The good thing is, we will run the numbers for you. We’ve worked with people across multiple industries who have requested gas purification for flow rates ranging from 50 mL/min to 50 L/min. Our sales team is well equipped to help you estimate O2 capacity and purifier life for off-nominal flow rates. For inquiries, please contact us at Sales@ChromRes.com.